WSW, NY, May 13th, 2026, FinanceWire
In 2012, Novo Nordisk (NYSE: NVO) had a once-weekly version of liraglutide, the molecule behind its diabetes drug Victoza, already in early-stage human trials. The trials had confirmed the drug was safe. Novo discontinued the program anyway. The company explained at the time that a separate molecule, semaglutide, had been assessed to have a more attractive profile for once-weekly use. Semaglutide went on to become Ozempic, Rybelsus, and Wegovy, the foundation of what is now one of the most valuable franchises in pharmaceuticals. Palatin Technologies (NYSE American: PTN) might have just made a similar move.
The small-cap biotech deliberately pushed back the timeline on the oral drug that has been the centerpiece of its obesity story, because it believes the next-generation molecule it is now advancing in its place is significantly better. The company had been targeting the first half of calendar 2026 to file an Investigational New Drug Application (IND), the regulatory step that allows human trials to begin, for PL7737, an oral pill designed to activate a brain receptor called MC4R that controls appetite and body weight. Palatin will now file that IND in the first half of 2027, but for a next-generation version of the molecule. The company says that the molecule, in preclinical models, shows materially improved receptor selectivity, with the potential to eliminate the hyperpigmentation associated with first-generation MC4R drugs.
In other words, Palatin is signaling it may be able to not just deliver an improved version of the drug that is on the market, but that it can maybe solve one of its largest drawbacks.
Vertex Pharmaceuticals (NASDAQ: VRTX) ran a similar play in pain. Its first-generation drug, VX-150, had produced positive mid-stage data in multiple pain conditions before Vertex set it aside for a next-generation molecule, suzetrigine, designed to hit the same target with a better profile. The FDA approved suzetrigine, marketed as JOURNAVX, in January 2025 as the first drug in a new class of non-opioid pain medicines in more than two decades. By mid-October 2025, more than 300,000 prescriptions had been written. The successor did what the first generation could not.
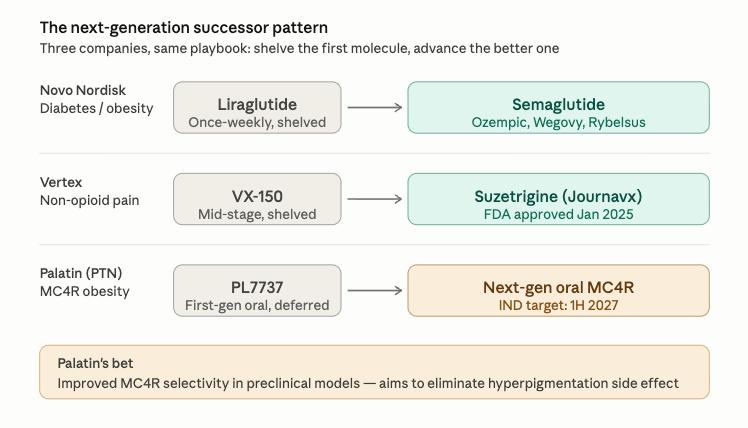 Palatin’s argument is that the MC4R class is now at a similar inflection point. The biology has been validated by Rhythm Pharmaceuticals (NASDAQ: RYTM), whose injectable MC4R drug IMCIVREE is approved and whose first-generation oral candidate Bivamelagon is advancing. What has not been solved is tolerability. Activating MC4R, the appetite receptor, can spill over onto a related receptor, MC1R, which controls skin pigmentation, producing a side effect called hyperpigmentation, or skin darkening. Selectivity, meaning a molecule that hits MC4R without also activating MC1R, is what the next generation of MC4R drugs is competing on, and what Palatin says its preclinical data shows. Whether that translates from animal models to humans will only be answered in the clinic, but the strategic logic mirrors what Novo and Vertex saw in their own programs.
Palatin’s argument is that the MC4R class is now at a similar inflection point. The biology has been validated by Rhythm Pharmaceuticals (NASDAQ: RYTM), whose injectable MC4R drug IMCIVREE is approved and whose first-generation oral candidate Bivamelagon is advancing. What has not been solved is tolerability. Activating MC4R, the appetite receptor, can spill over onto a related receptor, MC1R, which controls skin pigmentation, producing a side effect called hyperpigmentation, or skin darkening. Selectivity, meaning a molecule that hits MC4R without also activating MC1R, is what the next generation of MC4R drugs is competing on, and what Palatin says its preclinical data shows. Whether that translates from animal models to humans will only be answered in the clinic, but the strategic logic mirrors what Novo and Vertex saw in their own programs.
There is a second piece of the pipeline that anchors the near-term catalyst calendar. Palatin’s once-weekly injectable MC4R selective peptide agonist remains on track for an IND submission in the fourth quarter of calendar 2026, ahead of the oral next-generation candidate. That program, not the oral, is now the first clinical candidate likely to read out. Both programs target the same rare obesity conditions: hypothalamic obesity, Prader-Willi syndrome, and Bardet-Biedl syndrome. These are genetic or acquired forms of obesity where the MC4R pathway is broken, which is why GLP-1 drugs like Ozempic, which work upstream, do not solve the underlying problem in these patients. Palatin is building for the population the GLP-1 class does not adequately reach.
Palatin’s focus is the next-generation oral and injectable MC4R programs, but the company has something most small-cap biotechs at this stage do not: a back catalog of previously discovered drugs that it has out-licensed, now delivering tranches of non-dilutive funding from a top-tier pharmaceutical partner. The Boehringer Ingelheim partnership on retinal diseases delivered €7.5 million in upfront and research milestones in the second half of 2025. The sublicensing of dry eye program PL9643 to Altanispac Labs contributed $3.8 million in January 2026 and drove $3.9 million in collaboration and license revenue for the quarter, against zero in the prior-year period.
The MC4R pathway has already been commercially validated. Rhythm Pharmaceuticals’ IMCIVREE generated approximately $194 million in FY2025 net product revenue, up roughly 50% year over year, after a March 2026 FDA approval expanding its label into acquired hypothalamic obesity Palatin is targeting the same patient populations with what it argues could be a meaningfully better drug profile, on both selectivity and dosing form. The front-runner has the head start and the revenue; the challenger has the freedom to design for what the market still does not have. Palatin has just told the market which lane it chose to run in.
Read this Next >>After Makary: What the FDA’s Leadership Vacuum Means for Drug Development
Recent News Highlights from Palatin (AMEX:PTN):
Palatin Reports Fiscal Third Quarter 2026 Financial Results and Provides Business Update
Palatin Reports Second Quarter Fiscal Year 2026 Financial Results and Provides Corporate Update
Palatin Reports First Quarter Fiscal Year 2026 Financial Results and Provides Corporate Update
Important Disclaimers and Disclosures: The author, Wall Street Wire, is a content and media technology platform that connects the market with under-the-radar companies. The platform operates a network of industry-focused media channels spanning finance, biopharma, cyber, AI, and additional sectors, delivering insights on both broader market developments and emerging or overlooked companies. Wall Street Wire is not a broker-dealer or investment adviser. References to market size estimates, valuations, price targets, or other third-party data are provided strictly for informational purposes. Wall Street Wire receives cash compensation from Palatin Technologies, Inc. (the “Issuer”) for coverage and awareness services, which are provided on an ongoing subscription basis. The content above is a form of paid advertising and promotion and is for informational purposes only and does not constitute financial or investment advice. This article may contain forward-looking statements about the Issuer’s products, plans, or prospects that are subject to risks and uncertainties; actual results may differ materially, and readers should review the Issuer’s public filings on SEC EDGAR (sec.gov/edgar) for full risk factors. Market size figures, research estimates, or other third-party data referenced in this article are quoted from publicly available sources believed to be reliable; however, we do not independently verify or endorse them, and additional figures or estimates may exist. Full compensation details, information about the operator of Wall Street Wire, and the complete set of disclaimers and disclosures applicable to this content are available at: wallstwire.ai/disclosures. This article should not be considered an official communication of the Issuer.

