WSW, NY, May 11th, 2026, FinanceWire
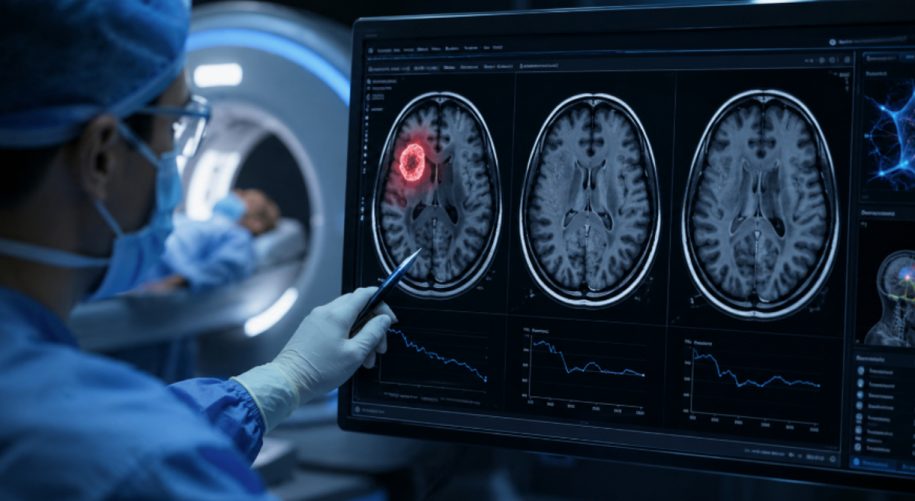
Glioblastoma is the most aggressive brain cancer in humans, and when it comes back after initial treatment, which it almost always does, the options run out fast. Surgery often is no longer safe. Median survival is measured in months. Across decades of clinical trials, the share of patients whose tumors visibly shrink on these salvage therapies has ranged from about 3% on biologic drugs to roughly 27% on the best antiangiogenic agents, according to a 2023 meta-analysis of 68 prior trials and nearly 4,800 patients. Against that baseline, the interim results Alpha Tau Medical (NASDAQ: DRTS) reported Monday morning are unusually strong, in the company’s own characterization, for a setting where complete responses to existing therapies are rare.
In groundbreaking interim data from its U.S. REGAIN trial, the company treated three patients with recurrent glioblastoma using a therapy called Alpha DaRT. Two of them had complete responses, meaning every visible tumor lesion disappeared on follow-up MRI scans. The third had stable disease with a 30% reduction in tumor size. That works out to a 100% local disease control rate, meaning every treated patient’s tumor either shrank or stopped growing, and a 67% complete response rate. Safety so far has been favorable in a procedure setting where seizures and transient neurological effects are well-known risks of any brain cancer intervention. The only serious adverse event was a seizure with temporary paralysis in one patient, which resolved with steroids. No unanticipated adverse events were reported, and no patient has shown local or distant disease recurrence at the May 3 data cutoff.
The caveat should travel with every number. This is an interim analysis of three patients, triggered by an FDA-requested safety review at a pre-specified milestone. The trial is designed to enroll up to ten U.S. patients, and early signals in tiny populations can fail to hold as enrollment grows. What makes the readout stand out is the size of the gap between the dataset and the historical baseline. In a disease where complete responses to existing therapies are vanishingly rare, two of the first three patients losing all visible tumor on MRI is a signal that warrants attention even at n=3.
Alpha Tau is the developer of Alpha DaRT, an alpha-radiation cancer therapy delivered directly into a tumor. Tiny radium-224 impregnated sources are placed inside the tumor, and as the radium decays, its short-lived daughters diffuse a short distance while emitting high-energy alpha particles designed to destroy nearby cancer cells while sparing surrounding healthy tissue. For brain cancer, that physics matters. Conventional external beam radiation can deliver only so much dose before damaging the sensitive brain tissue around the tumor. Alpha emitters travel a much shorter distance, allowing far higher local dose without comparable collateral injury. Principal Investigator Dr. Joshua Palmer at The Ohio State University Comprehensive Cancer Center described it as addressing “a long-standing problem in CNS radiation oncology.”
The delivery story is also new. To run REGAIN, Alpha Tau built an intracranial delivery approach that leverages the same stereotactic navigation neurosurgeons already use every day for routine brain biopsy. No new infrastructure, no new procedure type. Dr. J. Bradley Elder, Director of Neurosurgical Oncology at OSUCCC, noted that recurrent glioblastoma “sits at the limits of what conventional neurosurgery can offer,” and a minimally invasive workflow that can intervene precisely at that point is what the approach was designed to enable.
The commercial setting is meaningful. Roughly 14,000 Americans are newly diagnosed with glioblastoma each year, recurrence is virtually inevitable, and the five-year survival rate remains below 10%. The FDA granted Alpha DaRT a Breakthrough Device Designation in glioblastoma in 2021, one of three the platform now holds.
The readout also lands in an unusually dense stretch of news. Eight days earlier, at Digestive Disease Week 2026, Alpha Tau reported a 100% local disease control rate in 19 evaluable patients across two early-stage pancreatic cancer trials. The company said it will continue treating patients in REGAIN pending FDA confirmation following review of the safety report. With ASCO pancreatic data on May 21, the AHNS head and neck cancer podium presentation in July, and ReSTART skin cancer data maturing toward a complete PMA submission, the catalyst calendar through the back half of 2026 looks dense. For a company that has spent years building a multi-indication platform around a single physics principle, the first clinical signal from the hardest indication on its roadmap is now in hand.
Recent News Highlights from Alpha Tau Medical (DRTS):
Important Disclaimers and Disclosures: The author, Wall Street Wire, is a content and media technology platform that connects the market with under-the-radar companies. The platform operates a network of industry-focused media channels spanning finance, biopharma, cyber, AI, and additional sectors, delivering insights on both broader market developments and emerging or overlooked companies. The content above is a form of paid promotional content and advertising. Wall Street Wire receives cash compensation from Alpha Tau Medical Ltd or a related conflicted party for ongoing coverage and awareness services. This content is for informational purposes only and does not constitute financial or investment advice. Wall Street Wire is not a broker-dealer or investment adviser. Full compensation details, information about the operator of Wall Street Wire, and the complete set of disclaimers and disclosures applicable to this content are available at: http://wallstwire.ai/disclosures. Market size figures or other estimates referenced in this article are quoted from publicly available sources; we do not independently verify or endorse them, and additional figures or estimates may exist. This article should not be considered an official communication of the issuer.